THE EXCHANGE | Newsletter - Jul 2023
Irreversible Electroporation - A Novel Technology to Treat Cancer Patients Unfit for Surgery
A 57-year-old male suffering from chronic liver disease arising from non-alcoholic steatohepatitis (NASH) with features of portal hypertension manifesting in the form of gastric varices and hypersplenism presented at Medanta - Gurugram with an incidentally detected renal mass lesion in the right kidney. The lesion, detected during a routine annual dynamic CT Scan, was approximately 2.6cm in size, predominantly exophytic, closely abutting the midpolar calyx and had imaging characteristics of a renal cell carcinoma (RCC).
In view of multiple comorbidities, including chronic liver disease (CLD) and very low platelet count (<20,000) as a manifestation of hypersplenism secondary to portal hypertension, the patient was considered to be at high risk for surgery. He was advised to consult an interventional radiologist to explore a minimally invasive management strategy with the intention to treat the disease with minimal morbidity in the form of post-procedure recovery and hospital stay.
On reviewing the patient and his reports, we found that the lesion was less than 3cm in size, making the patient eligible for thermal ablation with radiofrequency ablation (RFA). However, due to the proximity of vital structures – the mid polar calyx in the vicinity of tentative ablative field – we advised to place a ureteric catheter in the upper ureter so that a continuous flush could be set in place to dissipate the heat in the calyx, and prevent or minimise the thermal trauma.
However, as RFA antennae constitutes a thicker needle (14G), there is always a risk of bleeding associated with it. This is an important factor to consider, especially in patients who consistently have a very low platelet count – a component responsible for initiating plug formation essential for formation of clots.
To minimise risk, the patient was advised large transfusion of platelets ahead of the procedure. The patient rejected this option owing to personal bias. Despite detailed counselling, he chose to get a transarterial embolisation done – a procedure that was offered as a relatively safer option in the absence of transfusion. Transarterial embolisation would control the disease by cutting off the tumour’s blood supply by selectively entering the blood vessels feeding the tumour through a small access from the femoral artery in the groin.
During follow-up after a month, the patient showed good response with no enhancing component.
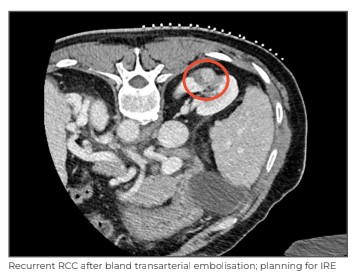
However, a follow-up scan after 6 months showed a 7mmx5mm enhancing nodule along the lateral aspect of the embolised lesion, suggestive of recurrence.
The patient was again advised ablation as the primary curative strategy. This time, instead of thermal ablation with RFA we offered the patient the newly launched technique of irreversible electroporation (IRE).
IRE is a newer tissue ablation technique in which micro-to-millisecond electrical pulses are delivered to produce cell necrosis through irreversible cell membrane permeabilisation. This also generates a local and systemic immune response to the tumour cells and destroys it. Crucially, it leaves the collagen-and elastin-rich extracellular matrix unaffected, aiding in minimising collateral damage of normal tissue.
This unique property of IRE used in the kidney spares vital structures, such as blood vessels and the renal collecting system.
IRE has an edge over thermal ablation in tumours like the one our patient had - the kind that lie very close to the renal calyces or pelvis wherein heat from thermal ablation may cause trauma to these structures. Moreover, the efficacy of thermal ablation also drops due to proximity of vessels leading to the “heat sink effect” that causes heat dissipation because of the flowing blood. These phenomenon are not seen with IRE.
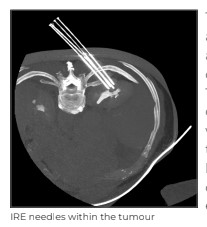
The procedure requires placement of 3-4, 19g needles around the lesion. These needles are relatively thinner compared to RFA / microwave antennae (MWA). By the virtue of being thinner, they are associated with lower incidence of bleeding – an important factor in our case. As the ablation zone is much more controlled with IRE, and because of its unique property of sparing blood vessels and connective tissue despite the proximity to the calyces, placement of the extremely cumbersome uretric catheter was also not necessary in our case.
The patient was again sensitised towards the need for pre-procedure platelet transfusion to make the procedure safer. With the risk being considerably lower than the previously offered RFA treatment, the patient consented.
The procedure was performed under general anaesthesia and 4 parallel needles were placed around the lesion under CT guidance to attain a tumour-free margin of 0.5cm. Eighty pulses were given through these needle combinations in sync with the patient’s heartbeat.
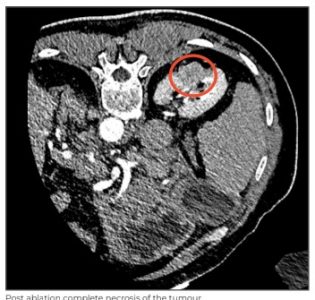
Post-procedure, on-table contrast CT showed no evidence of enhancement in the tumour and no evidence of bleeding was noted post procedure. The patient’s recovery in the hospital was uneventful and comfortable. The patient was discharged on Day 2 of the procedure. He has recovered well and has reported significant improvement in his quality of life.
In conclusion, IRE has emerged as a great ablative strategy for patients with multiple comorbidities, not fit for surgery. It has an edge over thermal ablative options for tumours proximal to vital structures and vessels; IRE gives similar oncological outcomes with fewer clinically significant adverse effects.
Irreversible Electroporation (IRE)
IRE, also known as Nanoknife, is a novel, non-thermal ablative therapy used to treat solid cancers. This treatment destroys cancer cells using electricity. It is applied by placing two or more needles in and around the tumour. Needles can be inserted percutaneously under image guidance, laparoscopically, or using an open surgical approach with intra-operative imaging.
Multiple short pulses of high-voltage electrical current are applied between needle pairs. The application of an electric field across cell membranes is thought to initiate the formation of nanopores in the lipid bilayer of the tumour cell’s plasma membrane, leading to homeostatic disruption and cell death.
Unlike other ablative techniques, such as MWA and RFA, IRE exert its cytotoxic effect without relying on thermal injury, sparing blood vessels and bile ducts. Furthermore, IRE is not susceptible to the “heat sink effect", a phenomenon where blood flowing in vessels adjacent to the cancer being treated prevents the area of ablation from reaching temperatures effective for cellular damage, leaving viable tumour cells , which is a concern with ablative methods reliant on thermal injury.
Dr. Sanjay Saran Baijal
Chairman - Diagnostic and
Interventional Radiology
Radiology and Imaging
Medanta - Gurugram
Medanta @ Work
Robotic Radical Nephrectomy with IVC Thrombectomy
Renal cell carcinoma is a condition that can have varied presentations ranging from being asymptomatic to pain in the loin or even blood in the urine. This cancer has a propensity to invade through adjacent vessels, such as the renal vein and inferior vena cava (IVC), as a tumour thrombus.
It is important to diagnose this condition early and treat it in time. Classically, patients with renal masses and venous thrombosis have been managed through open surgery as it is the most familiar technique and provides better control over the vessels. However, with the advent of robotic surgery, principles of open surgery can be replicated and combined with the advantages of minimally invasive surgery. Very few centres have reported the experience of performing robot-assisted radical nephrectomy with IVC thrombectomy. We report one such patient with right renal mass and IVC thrombus (Level 2) who underwent robotic right radical nephrectomy and IVC thrombectomy.
Case Study
A 65-year-old male presented at Medanta - Gurugram with the chief complaint of two episodes of haematuria (blood in urine) for 2 months. It was associated with the passage of long, thread-like clots. He had no other urological complaints. He had no addiction to smoking tobacco or any other substances. On examination, he was stable and had no evidence of pallor or any lymphadenopathy. The abdominal examination did not reveal any positive findings. Initially, he underwent an abdominal ultrasound at another centre that revealed a 65mmx50mmx65mm right renal mass. An abdominal contrast-enhanced computed tomogram (CECT) done elsewhere showed a right renal mass of size 88mmx65mmx103mm with a tumour thrombus infiltrating the right renal vein and IVC.
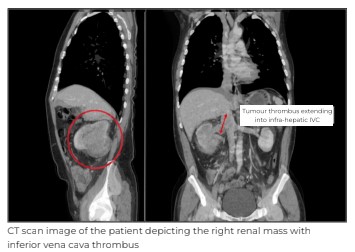
Further, whole-body FDG-PET (fluorodeoxyglucose-positron emission tomography) scan was done to confirm the absence of distant metastasis. His haemoglobin was 10.2gm/dL and creatinine was 1.2mg/dL. Other blood investigations were unremarkable. After a thorough discussion and imaging review, the patient was planned for robot-assisted radical nephrectomy with IVC thrombectomy.
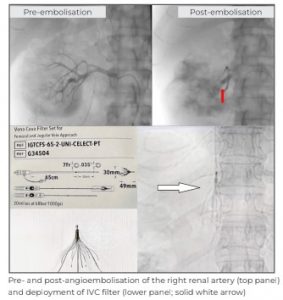
Preoperatively, we did a right renal artery angioembolisation with an IVC filter.
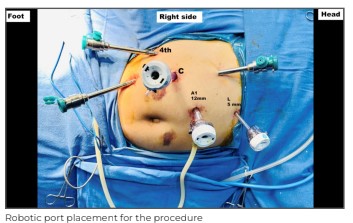
The patient was administered general anaesthesia and placed in a lateral position with 7 standard ports (4 robotic, 2mm-12mm assistant and 1mm-5mm assistant) placed for kidney docking. Robotic shears on right, fenestrated bipolar on the left and prograsp forceps in the fourth arm. Intra-operatively, we noted a large renal mass located in the mid and lower pole of the right kidney with dilated and enlarged right renal vein.
A tumour thrombus was seen extending up to 2cm into the IVC. The right kidney was mobilised beginning from the lower pole. The right ureter and gonadal vein were dissected and isolated. Then we performed the hilar dissection and the right renal artery was isolated and clipped. The kidney was again mobilised all around and left attached to the right renal vein. We then started IVC mobilisation from the infrarenal end. Infrarenal IVC was dissected and looped, followed by left renal vein and suprarenal IVC above the level of thrombus. Now, sequentially, lower IVC, left renal vein and suprarenal IVC were occluded by vascular loops and clamps.
The IVC was opened along the anterior border to deliver tumour thrombus and to remove the right renal mass.
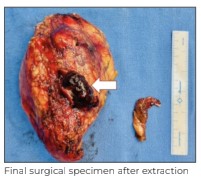
The IVC was closed in two layers using prolene 5.0. The vessel lumen was flushed with heparinised saline just before complete closure and vessel loops were released. After ensuring complete haemostasis and drain placement in the right renal fossa, we extracted the specimen from a pfannenstiel incision. Postoperative recovery was uneventful and the patient was discharged on Day 4. At a recent 3-month follow up, the patient was disease-free and doing well.
Expansion of minimally invasive surgery in this patient was associated with less pain, faster recovery and early discharge from the hospital. However, such complex procedures can be accomplished robotically due to their advantages over conventional laparoscopy, such as 3D vision, Endowrist and better precision.
Dr. Gagan Gautam
Vice Chairman - Uro Oncology and Robotic Surgery, Urology and Andrology
Kidney and Urology Institute
Medanta - Gurugram
Dr. Gopal Sharma
Consultant - Uro Oncology and Robotic Surgery, Urology and Andrology
Kidney and Urology Institute
Medanta - Gurugram
Complex Case of Pan Facial Fracture with Flail Nose
A 45-year-old male presented in the Emergency of Medanta - Ranchi with polytrauma. The patient was initially treated at another centre and referred to our hospital for further management.
On initial assessment, the patient was semiconscious and irritable. He had dyspnoea, pallor and tachycardia. There were signs of excessive bleeding from the nose and oral cavity as the nose and mouth were full of blood clots with gross laceration of the palate, nose and upper lip. There was considerable swelling and deformity of the face with malalignment of teeth, stitched wounds at the nose, upper lip and left eyebrow. The patient was breathing through the mouth as the nose was blocked with blood clots; the nasal bridge had collapsed completely, and was moving in and out with the patient’s breath - a condition known as a flail nose, just like a flail chest.
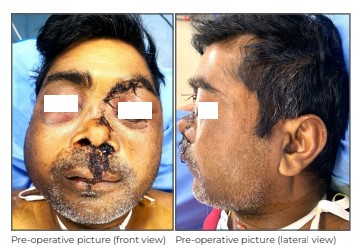
We immediately started resuscitating him and followed it up with basic investigations, including a CT
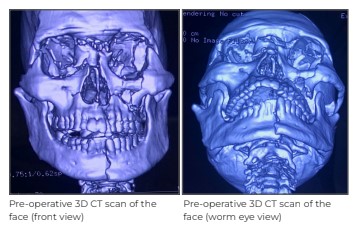
scan of the brain, face and chest. CT brain was suggestive of diffuse axonal injury; CT chest revealed
multiple rib fractures; CT face showed panfacial fracture, including a fractured mandible symphysis,
right temporo-mandibular (TM) joint dislocation, fracture of the hard palate, bilateral fronto- zygomatic and maxillary fractures along with comminuted depressed fracture of the nasal bones.
After initial assessment and investigations, the patient was shifted to the intensive care unit (ICU) in view of his critical condition. A tracheostomy was done to secure the airway and the patient was put on ventilator support. We continued his resuscitation, administered intravenous (IV) fluids, did blood transfusion and took other measures. The neurosurgeon and chest surgeon evaluated the patient, and conservative treatment was planned for his head and chest injuries.
Management of facial injury was the main challenge. Almost all facial bones were fractured with moderate to gross displacement. There was also soft tissue laceration of the face and palate requiring time-consuming surgical procedures. To avoid permanent disability and deformity of the face, early surgical intervention for facial injury was necessary. However, due to the associated life-threatening condition – mainly the brain injury - the facial surgery was deferred for 3 days till the patient’s condition stabilised and he got a go-ahead from the neurosurgeon.
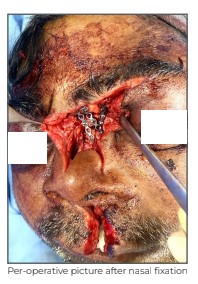
After 3 days, he was planned for surgical correction of the facial fractures. Close reduction of right TM joint was done, fracture symphysis of mandible was reduced with appropriate reduction forceps and clamps, and fixed with locking plates and screws. Bilateral fronto-zygomatic comminuted fractures were reduced and fixed with miniplates and screws. The fracture in the hard palate was reduced with maxillary disimpaction forceps and fixed with rectangular miniplate and screws; and the palatal laceration was repaired. The nasal fracture was approached through the existing wound on dorsum of the nose extending on left eyebrow. As the nasal bones were in multiple pieces, they were reduced by manipulation with nasal forceps and fixed with multiple miniplates and screws to achieve satisfactory nasal contour. The soft tissue of the nose and upper lip was repaired and maxillary fractures were taken care of using close reduction as they were relatively stable. Arch bars were applied to further stabilise the maxillary fractures, and to maintain satisfactory occlusion in
post-operative period with dynamic rubber band traction.
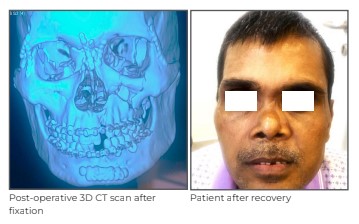
The 5-hour-long surgery was uneventful with good clinical outcome; satisfactory and stable fixation of facial fractures was obtained. The patient was on ventilator support for 4 days after the surgery, mainly due to brain and chest injury. He was discharged in a stable condition on Day 1 4 of admission.
Discussion
Fractures of individual facial bones are common but panfacial fractures pose a special challenge, more so when associated with other injuries that are life-threatening. They need special surgical expertise, skilled anaesthetic backup along with a dedicated critical care team, a neurosurgeon, a chest surgeon and other specialities to deal with associated injuries.
Nose is a conspicuous projection, and is more likely to be involved in any facial injury. Hence fracture of the nose is the most common among all facial bones. Most nasal fractures can be managed effectively by close reduction and splinting.
However, nasal fracture with flail nose is not common, and requires special attention and expertise as these fractures are unstable and reduction cannot be maintained without internal fixation. Internal fixation of flail nose itself is challenging as nasal bones are fractured into multiple small pieces that are unstable. It is mandatory to reduce the fractured segments, and to maintain the reduction till screws are drilled over miniplates. This is also difficult because of the size of the fractured fragments. Moreover, every case of panfacial fracture is different and poses unique challenges; the surgeon has to take decisions as per the situation.
Dr. Arvind Prakash
Associate Director - Aesthetic, Plastic and Reconstructive Surgery
Medanta - Ranchi
Welcome Onboard
 Dr. Balamurali Srinivasan
Dr. Balamurali Srinivasan
Associate Director - Cardiac Thoracic Surgery
Medanta - Ranchi
Cardiovascular and thoracic surgeon with over 16 years of experience and expertise in thoracic aortic surgery, repair and replacement of the heart valve, coronary artery bypass graft with focus on arterial grafts, minimally invasive surgeries, including MICS, VATS, and Mediastinal and Lung procedures.
 Dr. Shaleen Agrawal
Dr. Shaleen Agrawal
Senior Consultant - Radiation Oncology
Medanta - Lucknow
Radiation oncologist with expertise in volumetric modulated arc therapy (VMAT), stereotactic radiosurgery (SRS), stereotactic body radiotherapy (SBRT) and brachytherapy. He specialises in the treatment of head and neck cancers, breast cancer, brain tumour and gynaecological malignancies.
 Dr. Avinash Kumar
Dr. Avinash Kumar
Senior Consultant - GI Surgery, GI Oncology and Bariatric Surgery
Medanta - Patna
Gastrointestinal surgeon with expertise in advanced laparoscopic surgery, failed laparoscopy and open surgery, complex gastrointestinal surgery and bariatric surgery.
 Dr. Nishant Singh
Dr. Nishant Singh
Consultant - Orthopaedics
Medanta - Patna
Orthopaedic surgeon with expertise in knee ligament surgery, sports injuries, knee osteotomy, cartilage repair and regeneration, shoulder surgery, knee and shoulder arthroscopy in addition to knee and shoulder joint replacement surgery.
 Dr. Amulya Swati
Dr. Amulya Swati
Consultant - Obstetrics and Gynaecology
Medanta - Ranchi
Gynaecologist with expertise in managing high-risk pregnancy and delivery, infertility in addition to
adolescent health and perimenopausal health issues.
 Dr. Abhishek Shukla
Dr. Abhishek Shukla
Associate Consultant - Kidney and Urology Institute
Medanta - Indore
Urologist with expertise in performing minimally invasive surgeries of the kidney, ureteric calculi and prostate in addition to reconstructive urology, andrology, renal transplant and vascular access surgeries for chronic kidney disease. He also specialises in surgeries of cancers of the kidney, prostate, urinary bladder and adrenal gland.
 Dr. Ashish Sharma
Dr. Ashish Sharma
Associate Consultant - Interventional Cardiology
Medanta - Indore
Interventional cardiologist with expertise in coronary and peripheral angiographies, including
primary and complex angioplasties, implantation of temporary and permanent pacemaker in
addition to balloon mitral valvuloplasties, 2D and transesophageal echocardiograms.
 Dr. Rahul Dey
Dr. Rahul Dey
Associate Consultant - Paediatric Surgery
Medanta - Gurugram
Paediatric surgeon with expertise in paediatric and neonatal gastrointestinal surgery, urological
surgery, laparoscopic surgery, thoracic and video-assisted thoracic surgery.
 Dr. Tarique Naiyer Jamil
Dr. Tarique Naiyer Jamil
Associate Consultant - Internal Medicine
Medanta - Patna
Physician with expertise in treating infectious diseases, including malaria, tuberculosis and dengue.
He also specialises in managing cardiac and rheumatic diseases.
 Dr. Aparajita Kumar
Dr. Aparajita Kumar
Associate Consultant - Clinical and Preventive Cardiology
Medanta - Gurugram
Cardiologist with expertise in clinical and preventive cardiology, critical cardiology in addition to treadmill test, 2D echocardiogram, stress and dobutamine stress echocardiogram. She also specialises in the treatment of heart failure.