THE EXCHANGE | Newsletter September 2018
Success with End-stage Heart Failure
Heat failure mortality is known to be 50% in five years, much higher than most cancers. This case study is of a patient who persevered over 14 years and won over his failing heart. He was prescribed several treatment options to deal with heart failure at different stages of the disease. His commitment to treatment, adherence to therapy, and faith in the practitioner stand out in his disease journey, which finally concluded with a successful heart transplant.
This patient, a young, 24-year-old male was diagnosed with dilated cradiomyopathy in 2002 for which he had received a CRT device to tide over heart failure with left bundle branch block. In 2004, when he first saw us, his LVEF was 20%. A non-responder to CRT, he had severe LV dysfunction, and was symptomatic despite best medical management. He was NYHA class III / IV and was not tolerating any drugs including beta blockers. He was hypotensive and had significant mitral regurgitation.
In 2004, when heart transplants were not so mainstream and ventricular devices were unaffordable for majority, as an end- stage treatment we planned to repair his mitral valve with stem cell therapy. 180 ml bone marrow was aspirated from which mononuclear cell concentrate was extracted. This was injected into the myocardium at various points through direct intra-myocradial injections. Intra-operatively, he had substantial reduction of mitral regurgitation and eventually, did not require repair of mitral valve.
Post-operative, he responded very well to stem cell therapy. His condition improved consistently, and he became NYHA class I by the end of first year. His blood pressure improved and we could build him on beta blockers and ACE inhibitors in full dose. His weight was increasing, not because of fluid buildup, but improved appetite and his congestive heart failure issue being resolved. His walking distance improved from an initial 120 meters in 6 minutes to 500 meters in the same duration. LVED improved from 7.4 to 6.8; EF from 20% to 25%.
Whether this improvement was a result of stem cell therapy alone, or stem cells aiding the buildup of beta blockers and ace inhibitor therapy, which he was initially not tolerating, remains unclear. Whatever was the case, he experienced significant improvement.
For almost a decade, he did extremely well. He continued to be NYHA class I. His EF gradually improved to 30%, and he was able to tolerate beta blockers and ace inhibitors.
In 2012, he underwent a replacement of the CRT as it was detected to be end of life for the battery.
After completing 11 years of stem cell therapy, in 2015, he developed a respiratory infection which led to decompensated heart failure. Gradually, his tolerance for beta blockers reduced and the dose had to be tapered due to intolerance and worsening heart failure. While the diuretic therapy had to be built up, his EF was down to 20%, again. He was back in NYHA class III and was not even tolerating conventional drugs. Exercise tolerance test revealed that his VO2 max had dropped to 12.4 ml /kg / min from 15.4 ml /kg / min, earlier. It was now obvious that his one year outcome would be dismal. The only way of saving him was a heart transplant, the ultimate treatment option for end-stage heart failure.
We counseled him and his family, and encouraged them to register for a transplant. Meanwhile, his recurrent decompensating heart failure required frequent hospitalization for Pulse Milrinone therapy to improve his cardiac contractility and output. This, we hoped, would sustain him until he found a matching donor heart.
On 12 April, 2017, he was admitted for a transplant work up so that he could be registered for a heart transplant. His EF was down to 15% and he was NYHA class III / IV.
After the registration process was complete, and we were about to discharge him, while he was still in the ward, he developed ventricular tachycardia. He was immediately subjected to CPR and revived. In three days, he was extubated and safely shifted to the HDU.
Until he received a matching heart, the plan now was to upgrade the CRT to CRT-D. The procedure was planned for 4 May, 2017. A day prior to the procedure, he received a confirmation of a matching donor heart and instead of a planned CRT-D upgrade, he underwent a heart transplant. Successfully.
Having completed one year of post-transplant follow up, he is doing well, leading a high-quality life.
A true testament to the Medanta Spirit of Determined Optimism.

The Medanta Heart Failure Clinic monitored the case very closely while he received almost all possible treatments for heart failure over 14 years. The clinic is a channel of frequent, two-way communication between the hospital and patients like him, to closely track the condition and treatment outcomes.
Measuring High Foot Plantar Pressure Using the MatScan
Undue pressures at any one point on the plantar surface of the foot can not only precipitate skin breakdown but also interfere with its healing process. Elevated plantar pressures have been widely recognized as a causative factor in the development of several pedal pathologies, including diabetic foot ulceration.
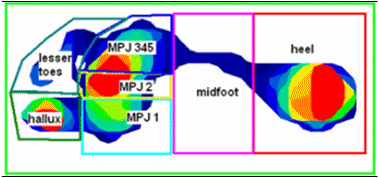
The analysis of plantar forces and pressures play a vital role in the healing of diabetic foot ulceration. Foot plantar pressure areas are measured by making the patient walk barefoot on a floor mat (Gait Analysis test) embedded with 2,288 sensing elements (sensors) and the pressure is measured in kPa. The pressure range is 125 psi/862 kPa. The sensing area is

Gait Analysis Test to determine areas prone to callus, corns, pain and ulceration
17.16 x 14.52" (435.9 x 368.8 mm), and the floor mat height is 0.225" (0.57 cm), making it extremely portable for maximum usage. This measurement system is valid as is documented in literature, confirming that it accurately quantifies dynamic plantar loading patterns of the foot. Maximum force (N), peak pressure (kPa) and average pressure (kPa) under the following regions of the foot are assessed: heel, mid-foot, 3rd-5th metatarsophalangeal joints (MPJ345), 2nd metatarsophalangeal joint (MPJ2), 1st metatarsophalangeal joint (MPJ1), hallux and the lesser toes.
Once these three variables are assessed, this information is gathered to manufacture re-distributive insoles that heal diabetic foot ulcer and further prevent their recurrence.
Medulloblastome - A Rare Childhood Cancer
Medulloblastoma is a malignant brain tumor that mainly occurs in childhood, accounting for approximately 20 percent of all primary tumors of the central nervous system among children less than 19 years of age. In India, its incidence is 22.4% of all the central nervous system tumors as reported from tertiary cancer care centers in the country.
What causes medulloblastoma?
The exact cause is unknown. Approximately 2 to 5 percent of medulloblastomas occur in association with either the nevoid basal cell carcinoma syndrome (NBCCS), caused by germline mutations in the patched-1 (PTCH1) gene, or familial adenomatous polyposis (FAP), caused by inactivating mutations in the adenomatous polyposis coli (APC) gene. Mutations in these genes predispose to the development of medulloblastoma through defect in pathways important in the pathogenesis of both sporadic and inherited tumors.
What are the common symptoms?
Major symptoms of medulloblastoma include increased intracranial pressure, cerebellar dysfunction (evolving over a period of weeks to a few months), gait ataxia or truncal instability, visual disturbance, cerebellar dysarthria (speech disturbance), dysdiadochokinesia (rapid repeated movements), tremors and limb clumsiness or incoordination. Other symptoms of cranial nerve involvement like numbness of face, difficulty in swallowing and reduced hearing are also noticed.
What are the different treatment options?
Combined modality therapy, including surgery, craniospinal radiation therapy and chemotherapy, is the standard of care for both children and adults with medulloblastoma.
Following maximum safe resection, the treatment of children with medulloblastoma varies according to the extent of disease and the age at diagnosis:
Average-risk disease: The average-risk sub-set includes children ≥3 years of age who have undergone a complete or near complete resection, have a negative cerebrospinal fluid cytology, and have no evidence of distant metastases. The treatment method for this category is combined modality treatment which includes both craniospinal RT +/- adjuvant combination chemotherapy.
High-risk disease: For this category, treatment method involves craniospinal RT with concurrent chemotherapy followed by combination chemotherapy.
Infants and young children: Children <3 years of age with medulloblastoma need reduced dose of craniospinal radiation for standard risk disease. Chemotherapy may be given to delay radiation, however, incidence of relapse/recurrence increases if radiation is not given.
Adults: Clinical experience with medulloblastoma in adults is limited, and treatment should be patterned on the same lines as in children. In adults with average-risk disease, combined modality treatment including both craniospinal RT and adjuvant combination chemotherapy is offered. The optimal treatment for adults with metastatic, unresectable, or recurrent medulloblastoma is unknown. Treatment includes craniospinal RT followed by combination chemotherapy rather than RT alone in most patients. In cases of recurrent medulloblastoma or patients with high-risk disease, gross residual post-surgery and with spinal metastasis, stem cell transplantation / bone marrow transplantation is recommended.
Are there any side-effects of the treatment?
There are several side effects that may occur when treating medulloblastoma: Posterior fossa syndrome, also called cerebellar mutism, is a major side effect which is characterized by impaired language production, inattention or difficulty initiating movement, cranial nerve palsies or bowel and bladder incontinence. Other side effects include neurocognitive impairment, hearing loss, short stature, endocrine abnormalities (growth hormone deficiency, thyroid deficiency, ACTH deficiency), cataract, cerebrovascular diseases, and even other secondary cancers.
Are people with specific genetic conditions more likely to develop the disease?
Approximately 2 to 5 percent of medulloblastomas occur in association with either the nevoid basal cell carcinoma syndrome (NBCCS), caused by germline mutations in the patched-1 (PTCH1) gene, or familial adenomatous polyposis (FAP), caused by inactivating mutations in the adenomatous polyposis coli (APC) gene. Mutations in these genes predispose to the development of medulloblastoma through defects in pathways important in the pathogenesis of both sporadic and inherited tumors.
What is the latest research on medulloblastoma?
Recent genomic studies indicate that medulloblastoma consists of at least four distinct molecular subgroups: WNT (wingless pathway), sonic hedgehog (SHH), Group 3, and Group 4. WNT and SHH medulloblastomas are characterized by aberrations in the WNT and SHH signaling pathways, respectively. WNT medulloblastoma has the best prognosis compared to the other medulloblastomas, while SHH medulloblastoma has an intermediate prognosis.
The underlying signaling pathways associated with Group 3 and 4 medulloblastomas have not been identified. Group 3 medulloblastoma is frequently associated with metastasis, resulting in a poor prognosis, while Group 4 is sometimes associated with metastasis and has an intermediate prognosis. Group 4 is the most frequent medulloblastoma and represents 35% of all medulloblastomas.*
(With inputs from Dr. Tejinder Kataria, Chairperson - Radiation Oncology, Cancer Institute, Medanta - The Medicity)
* Source: Northcolt . P.A., Buchhalter I. et al, The whole genome landscape of medulloblastoma subtypes. 20 Jul, 2017, Vol. 547: Nature . 311
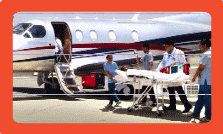
Medanta's Flying Doctors: An ICU at 30,000 Feet
Medanta is one of the few medical institutions in India to provide tertiary medical care on board an aircraft.
Medanta's Flying Doctors work to eliminate geographic barriers to enhance access to super specialized medical expertise and are available 24 hours a day, 365 days a year.
With its diverse fleet of aircraft, Medanta Air Ambulance is capable of both domestic and international medical evacuations.
Seamless patient transfers. How we do it?
- One call to get moving All calls to our air ambulance service number are attended by 24x7 on call doctors experienced in critical care. The doctor on call assesses the patient condition after evaluating case history, medical reports and clinical records.
- Preparing for evacuation Based on the assessment of the doctor, the appropriate medical team, equipment and aircraft are dispatched to pick-up the patient from any location, including remote areas with unmanned runways.
- Take-off At the Airport, we have the lowest activation time (time from receiving a call to take off) of 90 minutes. We also get priority for take-off and landing, including a green corridor during an emergency.
- On-board During flight, we are well-equipped and capable of providing advanced critical care support and can also conduct emergency procedures.
- There is complete ownership of the treatment provided to the patient by the Medanta Flying Doctors team.
- Airport to hospital Medanta runs the medical center at Delhi airport and can instantly evacuate the patient directly from aircraft door to Medanta - The Medicity in a seamless manner.
For query / booking, call Dr. Sunil Dubey on +91 9810736503
https://www.medanta.org/flying-doctor/
Emergencies happen without warning and they don't always happen at places where medical emergency services are accessible. In such conditions, medical air evacuation service acts like a lifesaver by making highly specialized emergency medical care accessible despite geographical challenges.
Dr. Naresh Trehan
CMD - Medanta